NGOs query GM mosquito success risk
(CNS) UPDATED 5 Oct, with Oxitec comment: Admissions by Oxitec, the UK company partnering with the Mosquito Research and Control Unit here in Cayman, that the space created by the successful eradication of the Aedes aegypti through the use of its genetically modified insects could pave the way for a second disease-carrying species has some international NGOs very concerned, but the firm has said the information is now out-dated. The watchdogs have said that the bio-engineered bugs create a risk that equally problematic mosquitoes will fill the vacuum created by the technology.
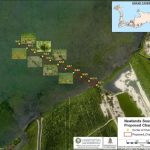
Referring to documents used during the recent case in the Cayman Islands Grand Court, when local activists challenged the decision to begin a test eradication project on a site in West Bay, the NGOs said that admissions by Oxitec show that the Aedes albopictus species, known as the Asian Tiger mosquito, which also transmits tropical diseases like dengue and Zika, could appear in the space left by the invasive Aedes aegypti if the genetically engineered insects are released in the United States.
“These court documents show that Oxitec’s GE mosquito trials are not worth the risk,” said the group, which includes GeneWatch, Friends Of The Earth, the International Centre for Technology Assessment and Food & Water Watch.
Oxitec states in the documents that if the Asian Tiger mosquito emerged to fill the space left by the Aedes aegypti, then a bio-engineered version of that species could address the issue. However, Food & Water Watch is pushing back and urging the authorities in Florida, who are set to engage in a project with Oxitec, not to release any genetically modified insects.
“It might be a good business model for a company to sell a technology to reduce one mosquito species, so then they can also sell a technology to deal with the species that replaces it,” said Wenonah Hauter, executive director of Food & Water Watch. “But it’s not worth the effort, expense and potential risk for communities in the US to start down this path.”
Aedes albopictus is a more invasive species than Aedes aegypti and can be found along the US east coast, from Maine to Florida, in all southern states, most midwestern states and in the states along the US-Mexico border, from Texas west to California.
“Current permits for releases should now be revoked until regulators recognise the downsides of Oxitec’s technology and the need to consider all the impacts on the ecosystem,” said Dr Helen Wallace, director of GeneWatch UK. “The consequences of mass releases of GE mosquitoes could be harmful if other disease-carrying mosquito species move in as a result. Risk assessments in Brazil, the Cayman Islands and the USA need to be revised.”
The NGOs have also accused Oxitec of hiding information about the GE mosquitoes’ potential to survive. Data has shown that 15-18% of its GE mosquitoes survive when fed on cat food containing industrially farmed chicken, which contains the antibiotic tetracycline. Environmental groups have warned that this could lead to the survival and spread of large numbers of GE mosquitoes when they encounter this common antibiotic in the environment in discarded takeaways or septic tanks.
“Oxitec has misled the public about the risks. These GE mosquitoes may thrive in the wild or may lead to an increase in more aggressive mosquito populations,” said Dana Perls, senior food and technology campaigner with Friends of the Earth, US. “We should be using the least toxic alternatives that don’t have unintended consequences for our environment and health.”
Florida Keys residents will have a non-binding vote on whether they support the release of Oxitec’s genetically engineered Aedes aegypti mosquitoes in November. Another vote will occur in Key Haven, Florida, where Oxitec has received permission from the Food and Drug Administration to release its GE Aedes aegypti mosquitoes in the first US trial. Residents of Key Haven have strongly opposed the release of these mosquitoes.
CNS contacted Oxitec Cayman and the MRCU about the latest concerns before publication but the following comment sent in the immediate wake of our enquiries was delayed.
“Oxitec has been and continues to be committed to transparency with policymakers, regulators and the public about its technology,” a spokesperson stated. “The question of whether the Aedes albopictus population might increase if the Aedes aegypti population decreased is something we have studied in previous projects including our year-long trial of Oxitec’s mosquitoes in Panama. That trial found no evidence that suppressing Aedes aegypti increased numbers of Aedes albopictus, and the data has been published in this peer-reviewed journal”
The firm added that the FDA-led review team including members of the CDC and EPA examined this concern for the Florida trial and found it was highly unlikely that the release of OX513A mosquitoes would lead to an increase in the population of other mosquito species that might contribute to an increase in disease transmission.
The information referred to by the NGOs comes from an outdated permit application which is no longer valid, Oxitec added. “It has been superseded by a more recent permit issued under the newly enacted Cayman Conservation Law whereby the government, under the direction of the National Conservation Council, has not mandated monitoring for Aedes albopictus. The outdated material refers to a potential academic research collaboration on Aedes albopictus that was under consideration at the time of application. The current permit best reflects the evolving priorities of the MRCU, and does not include an element of Aedes albopictus control using the Oxitec technology,” the bio engineering firm told CNS.
“We do a level of Aedes albopictus monitoring that we consider relevant to the project and consistent with local conditions, but this is not a condition of the regulatory permit…it is also important to note that Aedes albopictus is not as good a vector as Aedes aegypti and that the transmission would not be increased by the suppression of Aedes aegypti,” the spokesperson added.
Category: Health, Land Habitat, Medical Health, Science & Nature


































Is it gospel that as long as the zika carrying mosquitoes are present, no other mosquito will move in to carry zika? And when (and if) our present day zika carrying mosquitoes are genetically engineered out of existence, another zika carrier will move in? I don’t believe that! Has it been proved that the current zika transmitter prevents another species from moving in? I thought not! I wonder how it would be to have NO mosquitoes……
The company inovio pharma is currently trying out a vaccine against zika this vaccine looks promising. has any one in government even look at that? or try to find out more about it? we need to look at all options out there
when your wife, mother, granddaughter, daughter, or you have a deformed child who will you blame?
You argue about gmo mosquitos but then turn around and eat gmo foods right after this article, how unintelligent!
No special interests here, eh?
Yes, let’s not try anything new or science based cuz the current chemical treatments are working so well. The leukemia doesn’t show up for a couple decades so lets allow our kids to swim in it.
These two mosquitoes species are radically different in terms of their rate of successful adaptation to human habitation. Among the 36 known species of mosquito in the Cayman Islands, Aedes albopictus has already been resident in “relatively low numbers” since it arrived in Grand Cayman in 1997 and has not proliferated to the extent that the much more recently arrived and uniquely adapted Aedes Aegypti has. If the Aedes Aegypti were to be successfully obliterated, a few isolated pockets of struggling and treatable Aedes Albopictus would be a wonderful problem for MRCU to have. Let’s hope we have those kind of problems!
Admissions by Oxitec, the UK company partnering with the Mosquito Research and Control Unit here in Cayman, that the space created by the successful eradication of the Aedes aegypti through the use of its genetically modified insects could pave the way for a second disease-carrying species has some international NGOs very concerned. The watchdogs have said that the bio-engineered bugs create a risk that equally problematic mosquitoes will fill the vacuum created by Oxitec’s technology.
People the above statement should cause every man jack in Cayman to come out and tar and feather and cowitch the backside of those stalwart proponents of OXITICK an its bush science. Oh people cant you see they took advantage of our ignorance and our usual “Foreigner knows best” fraking attitude, So tired of the BS in this country where Caymanians wont respect each other and allow every johny come lately or even living her for a long time to blind us from the truth. As Dwayne and the petition tried to invoke this bombo C testing that they did was not a sure ting XXXX.
People learn that we have our own who do not want to hurt us for in so doing they hurt themselves. Knowledge is power and there are many caymanian’s with Knowledge, you just have to luw them to speak. Listen people and you will hear and you will learn stop the bull crap chatter . Listen and learn then elect those who indeed mean well and have the requisite knowledge to run this ya country.
So your solution to the possibility that this solution may not be a permanent solution is to not try the solution to see if it really works or not? There are few absolutes in life, neighbour. Death, taxes, Jesus. The first two won’t help here and the third helps those who help themselves. (Go MRCU.)
Thank you for posting this article I was worried it would be hidden
“Oxitec has misled the public about the risks” says it all, I wonder if had the judge known about the all the truth as it will continue surface, if he would of granted the release
The judge knew. As the article states, this ‘objection’ (to the Florida release) is sighting information disclosed in the Cayman case (because being able to say “Referring to documents used during the recent case in the Cayman Islands Grand Court” is so much more exciting copy that saying ‘referring again to an already well known and addressed possibility’) so the Judge knew about it. And with that knowledge SHE granted the approval for the release to continue.
So their objection is ‘don’t kill the bad alien mosquito because another bad alien mosquito MIGHT take its place’. – Not very convincing.
There are going to be a lot more “admissions” by Oxitec in the near future I’m sure. Yes, science has taken amazing strides to better human life on this planet – but modifying nature genetically is a slap in Mother Nature’s face and believe me it will ALWAYS have adverse consequences
Mother Nature is the ultimate genetic modifier and like justice she is blind.
It is always easy to come in and criticize, but unless you can come up with an alternate solution, it carries no weight.